
Sara Vitalini 1, 5,*, Stefania
Garzoli 2, Valentina Vaglia3 and
Marcello Iriti44, 5,*
Received: 2022-07-07 | Revised:2022-07-19 | Accepted: 2022-08-08 | Published: 2022-08-19
Abstract
Weeding is a practice of primary importance both in
conventional agriculture where the intensive use of herbicides can represent a
significant environmental risk and in organic farming where this practice is
prohibited. Therefore, the need to identify alternative means of controlling
weeds is evident. In this respect, allelopathy proves to be a useful tool to be
integrated with conventional agronomic techniques for the management of
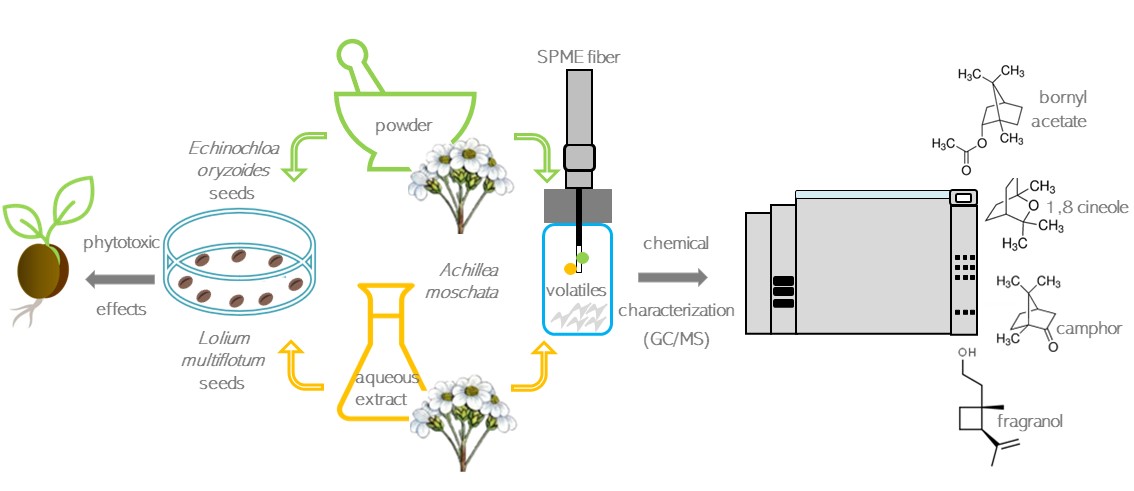
infesting flora. In this work, we investigated the phytotoxic potential of Achillea
moschata Wulfen (Asteraceae) against Echinochloa oryzoides (Ard.)
Fritsch and Lolium multiflorum Lam., two noxious plants for crops
such as rice and wheat. Preliminary anti-germination assays were carried out in
controlled conditions by
testing both the powder (0.25 g) and the aqueous
extract (1%, 10% and 20%) from the A. moschata aerial parts. The obtained
results showed that the powder was more effective than the extract in
inhibiting seed germination (up to –81.6% vs –48.8% at 20% concentration) and
seedling development (up to –99.4% vs –75.9% at 20% concentration) of both
target species, although L. multiflorum was more susceptible than E.
oryzoides to treatments. Furthermore, the chemical composition of the two
used matrices was characterized by Solid-Phase
Microextraction (SPME) sampling technique and Gas Chromatograph/Mass
spectrometer (GC/MS) analyses. Camphor (25.8% and 49.9%) and 1,8 cineole (25.9%
and 20.7%) were the main constituents in the samples, followed by bornyl
acetate (6.7%) in the powder and fragranol (10.5%) in the aqueous extract.
Keywords
Achillea genus;
anti-germination activity; biocontrol; phytotoxicity; weed management; SPME-GC-MS
References
1.
Li Z.R.; Amist N.; Bai L.Y.
Allelopathy in sustainable weeds management. Allelopathy
J. 2019, 48, 109-138.
2.
Cai X.; Gu M. Bioherbicides in
organic horticulture. Horticulturae 2016, 2, 3.
3.
Godlewska, K.; Ronga, D.;
Michalak, I. Plant extracts - importance in sustainable agriculture. Ital. J. Agron. 2021, 16.
4.
Vitalini, S.; Orlando, F.;
Palmioli, A.; Alali, S.; Airoldi, C.; De Noni, I.; Vaglia, V.; Bocchi, S.;
Iriti, M. Different phytotoxic effect of Lolium
multiflorum Lam. leaves against Echinochloa
oryzoides (Ard.) Fritsch and Oryza
sativa L. Environ. Sci. Pollut.
Res. 2020, 27, 33204–33214.
5.
Vitalini, S.; Iriti M.; Orlanso,
F.; Garzoli, S. Chemical volatile composition and phytotoxic potential of Daphne gnidium L. leaves. Sustain. Chem. Pharm. 2022, 25,
100607.
6.
Pignatti, S. Flora d’Italia, Edagricole, Bologna; Vol. 3:
p.79; 1982.
7.
Al-Mudaris, M. Notes on various
parameters recording the speed of seed germination’. Tropenlandwirt 1998, 99,
147–154.
8.
Ellis, R.A.; Roberts, E.H. The
quantification of ageing and survival in orthodox seed. Seed Sci. Technol. 1981, 9, 373–409.
9.
Abdul-Baki, A.A.; Anderson, J.D.
Vigour determination in soybean seed by multiple criteria. Crop Sci. 1973, 1, 630–633.
10.
Garzoli, S.; Orlando, F.; Iriti,
M.; Vtalini, S. Solanum linnaeanum leaves:
Chemical profiling of VOCs and effects on seed germination and early growth of
monocots and dicots. Chem Biodivers. 2022, 19,
e202100975.
11.
Vitalini, S.; Orlando, F.; Vaglia,
V.; Bocchi, S.; Iriti, M. Potential role of Lolium
multiflorum Lam. in the management of rice weeds. Plants 2020, 9, 324.
12.
Polatoğlu, K.; Karakoç, Ö.C.;
Görenc, N. Phytotoxic, DPPH scavenging, insecticidal activities and essential
oil composition of Achillea vermicularis, A.
teretifolia and proposed chemotypes of A.
biebersteinii (Asteraceae). Ind.
Crops. Prod. 2013, 51, 35–45.
13.
Çakır, A.; Özer, H.; Aydın, T.;
Kordali, Ş.; Çavuşoglu, A.T.; Akçin, T., Mete, E.; Akçin, A. Phytotoxic and
insecticidal properties of essential oils and extracts of four Achillea species. Rec. Nat. Prod. 2016, 10, 154-167.
14.
Elshamy, A.; Abd-ElGawad, A.; Mohamed,
T.; El Gendy, A.E.; Abd El Aty, A.A.; Saleh, I.; Moustafa, M.F.; Hussien, T.A.;
Pare, P.W.; Hegazy, M. Extraction development for antimicrobial and phytotoxic
essential oils from Asteraceae species: Achillea
fragrantissima, Artemisia judaica and Tanacetum sinaicum. Flavour Fragr. J. 2021, 36, 352–364.
15.
Hatata, M.M.; El-Darier, S.M.
Allelopathic potential of an aromatic plant; Achillea
santolina L. on germination of Triticum
aestivum L. Egypt. J. Exp. Biol. (Bot.) 2009,
5, 131-141.
16.
Tammam, A.A.; El-Bakatoushi, R.;
El-Darier, S.M. The phytotoxic potential of Achillea
santolina L. (Asteraceae) on Vicia faba
L. and Hordeum vulgare L. Asia Life Sci. 2011, 20, 443-464.
17.
Pour, A.P.; Farahbakhsh, H.
Allelopathic effect of Achillea (Achillea santolina) on germination and
growth of crop plants. J. Agric. Sci.
Technol. 2011, 1045-1053.
18.
Darier, S.M.; Tammam, A.A.
Potentially phytotoxic effect of aqueous extract of Achillea
santolina induced oxidative stress on Vicia
faba and Hordeum vulgare. Rom. J. Biol. -Plant Biol. 2012, 57,
3-25.
19.
Vitalini, S.; Madeo, M.; Tava, A.;
Iriti, M.; Vallone L.; Avato, P.; Cocuzza C.E.; Simonetti, P.; Argentieri,
M.P. Chemical Profile, Antioxidant and Antibacterial Activities of Achillea moschata Wulfen, an Endemic
Species from the Alps. Molecules 2016, 21,
830.
20.
Tava, A.; Iriti, M.; Vitalini,
S. Composition and antioxidant activity of the essential oil from Achillea moschata Wulfen growing in
Valchiavenna and Valmalenco (Italian Central Alps Int. J. Hortic. Sci. Technol. 2020, 7,
335-341.
21.
Argentieri, M.P.; Madeo, M.;
Avato, P.; Iriti, M.; Vitalini, S. Polyphenol content and bioactivity of Achillea moschata from the Italian and
Swiss Alps. Z. Naturforsch. 2020, 75c,
57-64.
22.
Chandler, R.F.; Hooper, S.N.;
Harvey, M.J. Ethnobotany and phytochemistry of yarrow, Achillea millefolium, Compositae. Economic Bot. 1982, 36, 203–223.
23.
Ali, S.I.; Gopalakrishnan, B.;
Venkatesalu, V. Pharmacognosy, phytochemistry and pharmacological properties of Achillea millefolium L.: A review. Phytother. Res. 2017, 31, 1140–1161.
24.
Agar, O.T.; Dikmen, M.; Ozturk,
N.; Yilmaz, M.; Temel, H.; Turkmenoglu, F. Comparative studies on phenolic
composition, antioxidant, wound healing and cytotoxic activities of selected Achillea L. species growing in Turkey. Molecules 2015, 20, 17976–18000.
25.
Venditti, A.; Guarcini, L.;
Bianco, A.; Rosselli, S.; Bruno, M.; Senatore, F. Phytochemical analysis of Achillea ligustica All. from Lipari
Island (Aeolian Islands). Nat. Prod. Res. 2016, 30,
912–919.
26.
Zengin, G.; Aktumsek, A.; Ceylan,
R.; Uysal, S.; Mocan, A.; Guler, G.O.; Mahomoodally, M.F.; Glamočlija, J.;
Ćirić, A.; Soković, M. Shedding light on the biological and chemical
fingerprints of three Achillea species
(A. biebersteinii, A. millefolium and A. teretifolia). Food
Funct. 2017, 8, 1152–1165.
27.
Afshari, M.; Rahimmalek, M.;
Miroliaei, M. Variation in polyphenolic profiles, antioxidant and antimicrobial
activity of different Achillea species
as natural sources of antiglycative compounds. Chem.
Biodivers. 2018, 15, e1800075.
28.
Yilmaz, M.A.; Ertas, A.; Yener,
I.; Akdeniz, M.; Cakir, O.; Altun, M.; Demirtas, I.; Boga, M.; Temel, H. A
comprehensive LC-MS/MS method validation for the quantitative investigation of
37 fingerprint phytochemicals in Achillea species:
A detailed examination of A. coarctata and A. monocephala. J.
Pharm. Biomed. Anal. 2018, 154, 413–424.
29.
Lee, H.J.; Sim, M.O.; Woo, K.W.;
Jeong, D.-E.; Jung, H.K.; An, B.; Cho, H.W. Antioxidant and antimelanogenic
activities of compounds isolated from the aerial parts of Achillea alpina L. Chem. Biodivers. 2019, 16, e1900033.
30.
Dehsheikh, A.B.; Sourestani, M.M.;
Dehsheikh, P.B.; Mottaghipisheh, J.; Vitalini, S.; Iriti, M. Monoterpenes: Essential
oil components with valuable features. Mini
Rev. Med. Chem. 2020, 20, 958–974.
31.
Okamoto, Y.; Yamaji, K.;
Kobayashi, K. Allelopathic activity of camphor released from camphor tree (Cinnamomum camphora). Allelopathy J. 2011, 27, 123–132.
32.
Nishida, N.; Tamotsu, S.; Nagata,
N.; Saito, C.; Sakai, A. Allelopathic effects of volatile monoterpenoids
produced by Salvia leucophylla:
Inhibition of cell proliferation and DNA synthesis in the root apical meristem
of Brassica campestris seedlingsJ. Chem. Ecol. 2005, 31, 1187–1203.
This work is licensed under the Creative Commons Attribution 4.0 License (CC BY-NC 4.0).