
Agus Limanto, Adelina Simamora and Kris
Herawan Timotius*
Received: 2023-03-08 | Revised:2023-04-13 | Accepted: 2023-04-15 | Published: 2023-05-23
1. Introduction
Xanthorrhizol (XNT) (2-methyl-5-[(2R)-6-methylhept-5-en-2-yl]
phenol) is a bisabolane-type
sesquiterpenoid (Fig. 1). It can be extracted from the rhizome of Curcuma xanthorrhiza (local name:
temulawak). The rhizome is an essential component of Indonesian
traditional medicine (local name:Jamu) and is extensively utilized as a
medicinal and nutritional plant. Traditionally, it is used to treat diseases
like lack of appetite, children's fever, stomach and liver disorders,
constipation, bloody diarrhea, dysentery, arthritis, hypotriglyceridaemia [1], hemorrhoids, and
rheumatism [2]. It also has various bioactivities, like
antioxidant, anti-inflammatory, anticancer, antidiabetic, antihypertensive, antiplatelet,
antimicrobial, skincare, and nephron-hepatoprotective properties [1-4]. Our body has appetite-related hormones (orexigenic
hormones), like ghrelin. As an orexigenic hormone, ghrelin (acylated ghrelin,
AG) regulates homeostatic and reward-related feeding behavior. This acylated ghrelin
comes from the acylation of ghrelin from des acyl ghrelin (DAG). Ghrelin O-acyl
transferase (GOAT), a gut enzyme,
catalyzes the acylation of DAG [5]. In most cases, AG carries the octanoyl
group in its third amino acid, serine (Ser2) [6]. Ghrelin impacts hunger and metabolic regulation by binding
the ghrelin receptor (GHS-R) for signal activation [7]. By
signal activation of GHS-R, GHS-R can regulate energy homeostasis and body
weight. Ghrelin activation to GHS-R can directly stimulate appetite and
hunger signaling [7]. GHS-Rs are most highly expressed in the hypothalamus, distinctively the ventromedial
nucleus and arcuate nuclei. However, expression of GHS-Rs also
happens in other areas of the brain, including the hippocampus, substantia nigra, and ventral tegmental area. Outside the central nervous system,
GHS-Rs also exist in the heart, liver, and skeletal
muscle [6].
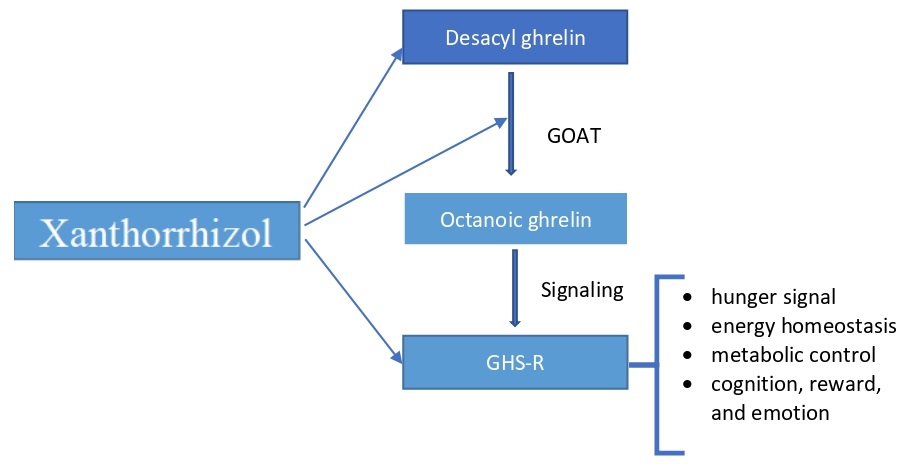
To
understand the relationship between ghrelin and appetite-related behavior, we
must know the structures and functions of ghrelin (AG), DAG, GOAT, and GHS-R.
Together is recognized as a Ghrelin/GOAT/GHS-R1a
system (G3S). Specifically, G3S is essential in energy homeostasis that
signals appetite and hunger [7].
Figure 1. Chemical structure of Xanthorrhizol
(PubChem)
2. Materials and methods
Can XNZ improve
appetite behavior by interacting with Ghrelin, GOAT, and GHS-R? What are the
molecular interactions between XNZ with Ghrelin, GOAT, and GHS-R? Can XNZ influence the activity
of GOAT and GHS-R signaling? and finally, can
XNZ modulate appetite behavior through its Interaction with GOAT and GHS-R?
We hypothesized that
the XNT has molecular interaction with Ghrelin and GOAT. In addition, XNZ has
strong interaction in the active or allosteric sites of GOAT and GHS-R.
Moreover, XNZ is an agonist of
GHS-R.
|
Receptors |
:
Desacyl Ghrelin (DAG), GOAT and GHS-R |
|
Endogenous
ligands of GOAT |
:
Desacyl Ghrelin (DAG) and o-octanoic acid |
|
Endogenous
ligand of GHS-R |
:
Ghrelin (acyl ghrelin, AG) |
|
Exogenous
ligands |
:
Xanthorrhizol |
3. Results and Discussion
3.1.
Octanoylation/Acylation process by GOAT
3.1.1. General
characteristics of ghrelin
Ghrelin is an
unusual peptide stomach hormone that is consisted of 28 amino acid residues.
Its desacyl form, DAG, undergoes acylation or octanoylation of its third amino
acid or Ser2. This acylated ghrelin (AG) is essential for ghrelin's activity to
signal its receptor, named GHS-R (Fig. 2). [8] GOAT catalyzes DAG octanoylation [7]. The produced ghrelin acts as an
endogenous ligand of GHS-R [8]. Therefore, DAG's acylation or octanoylation is essential for
releasing ghrelin-induced growth hormone from the pituitary that stimulates
appetite [9]. Ghrelin
is considered the only peripheral hormone to transmit satiety or appetite
signals. Nevertheless, ghrelin has additional physiological functions, like the
stimulation of growth hormone release and accumulation of fat (obesity) (Fig.
2) [8, 10].
Figure 2. The function of ghrelin as a
controller of homeostatic and hedonic feeding
Ghrelin is one of three hormone peptides encoded by the same
preproghrelin gene. The other two hormone peptides are DAG and obestatin (Fig.
3). They modulate appetite, adipogenesis, glucose metabolism, immunity, sleep,
anxiety, stress, and regulation of feeding-stimulated gastroduodenal motility.
The stomach may regulate gastrointestinal motility via AG, DAG, and obestatin [11]. Even ghrelin is produced in the
stomach, but its activities exert in the central nervous system by crossing the
BBB. The produced ghrelin can stimulate the secretion of growth hormone
(GH).
Figure
3. Three hormone peptides from a single
gene
Therefore, ghrelin is thought to directly affect neurons
involved in feeding via GH secretion by activated GHS-R (ghrelin receptor). Several
gastrointestinal hormones, including ghreline, can transmit signals to the
brain via the vagal afferent system. Vagotomy abolishes or attenuates GH
secretion and the ghrelin's action on feeding. The vagal
afferent system can convey the ghrelin's signals for feeding and GH
secretion to the brain [12]. Blood-brain
barrier (BBB) controls the entry of ghrelin, into the brain. Once ghrelin is
present in the brain, it can activate the hypothalamus for regulating food
intake, in the hippocampus for regulating neurogenesis, and in the olfactory
bulb for regulating food-seeking behavior [13].
A preproghrelin gene encodes three peptides, namely ghrelin
(or acyl ghrelin; AG), des-acyl ghrelin (DAG), and obestatin. Although DAG is
considered as a degradation product of AG, DAG is considered as a separate
hormone that has its own receptor and also can interact with AG at its
receptor. Actually, DAG is a functional inhibitor of AG [14].
AG, DAG, and obestatin are both active hormones [15]. They
are derived from a common prohormone, preproghrelin [16].
Ghrelin has orexigenic , but DAG and obestatin have anorexigenic properties.
Ghrelin is produced mainly in the stomach and is an endogenous ligand of GHS-R
located in the brain. The ghrelin levels in plasma strictly depend on recent
food intake. Therefore, it is essential in appetite and meal initiation [17]. While
ghrelin activates GHS-R, DAG does not [18]. Ghrelin
can activate the pituitary and hypothalamus in stimulating appetite and
adiposity and releasing growth hormone through its activation of GHS-R-1a.
However, DAG, the unacylated ghrelin form, does not bind GHS-R-1a and is devoid
of endocrine activity. But in plasma, DAG is more abundant than ghrelin [19].
3.1.2.General characteristic
of Ghrelin O-acyltransferase (GOAT)
The only peptide known to undergo
octanoylation is ghrelin. This octanylation is catalyzed by ghrelin
O-acyltransferase (GOAT). GOAT is able to attach octanoate to DAG, and then
produce AG, [20]. GOAT is expressed mainly in the
gastrointestinal (GI) tract [21], is secreted by stomach X/A-like cells,
and plays a role in appetite and metabolism [22]. DAG in the blood can cross BBB but it
cannot bind to GHS-R1a. AG, but not DAG, can upregulate the GOAT expression [23]. The presence of GOAT in the hippocampus
is essential for acylating DAG locally. The expression of GHS-R1a may be
related to the synthesis of GOAT in the hippocampus [24].
3.1.3.
Octanoylation process
GOAT
is the only recognized enzyme that can catalyze the acyl modification of DAG
that results in acylated ghrelin (AG). GOAT modifies the third amino acid
serine (Ser2), not the other DAG peptides' residues. DAG and n-octanoic acids
are substrates and ligands, respectively, for GOAT [25]. Octanoyl
acyl donor should be supplied externally. Additionally, a four-amino acid
peptide derived from the N-terminal sequence of ghrelin constitutes the core
motif for substrate recognition by GOAT [26]. GOAT esterifies an n-octanoic acid to DAG, resulting in acylated
ghrelin (AG) that can bind and activate the GHS-R (Fig. 2) [27, 28].
Ghrelin
has a vital role in regulating glucose
metabolism. GOAT can modify ghrelin into its active form [6, 29-31]. Its activity is associated with hedonic
feeding behavior that is mediated by forebrain orexin signaling. The
GOAT-ghrelin system is essential in mediating food motivation and hedonic
feeding [5].
Activation
or inhibition of GOAT depends on the physiologic situation.
The fasting and satiation conditions can activate GOAT. For the GOAT’s
activity, octanoic acid is needed as its substrate. GOAT can use octanoic acid
either from diet-derived or adipose-fatty acids. Dietary fatty acids are
probably a primary source of octanoate available in the stomach. However,
there is a possibility of endogenous production of octanoate in the
GOAT-expressing cells.
Moreover, the white adipose tissue can release fatty acids for GOAT to activate ghrelin, particularly during fasting. This situation is consistent with circulating ghrelin levels that increase during food deprivation. Long-term fasting can inhibit acylation but not the secretion of ghrelin. This situation is correlated with the ghrelin level that increases before meals and decreases after meals [32].
Inhibitors
of GOAT can indirectly decrease ghrelin levels [33]. Specific GOAT inhibitors of GOAT can
block an octanoyl attachment to ghrelin. GOAT is subjected to end-product
inhibition [21]. There are two groups of GOAT inhibitors:
ghrelin peptide-mimetic and small-molecule inhibitors (non-peptide-based GOAT
inhibitors). An example of a ghrelin mimetic inhibitor is GO-CoA-Tat, a kind of
peptide that antagonizes GOAT [30, 34]. GO-CoA-Tat
attenuates AG production and prevents weight gain. In addition, GO-CoA-Tat can
also increase glucose-induced insulin secretion. Therefore, inhibition of GOAT
is an alternative strategy for treating obesity and related metabolic disorders
[35]
Small-molecule
GOAT inhibitors, like triterpenoid GOAT inhibitors, compound A and B. Synthetic
triterpenoids are discovered and identified as CDDO
(2-cyano-2,12-dioxoleane-1,9(11)-dien-28-oic acid). Compound A
(2-[(2,4-dichlorobenzyl) sulfanyl]-1,3-benzoxazole-5-carboxylic acid) and
compound B (4-chloro-6- -1-benzothiophen-3-yl) acetic acid) can be synthesized
and inhibit GOAT. They show octanoyl-CoA competitive inhibitory activity and
can decrease acyl ghrelin production [22].
Ghrelin
is a potent food intake stimulator, leading to weight gain and adiposity. It
can increase the risk of obesity and binge eating behavior. The functionality
of ghrelin is due to its interaction with the GHS-R1a. Besides its ability to
promote the reinforcement of hedonic food, it also acts at extra-hypothalamic
sites, making interaction with dopaminergic, cannabinoid, opioid, and orexin
signaling [36].
3.2. Growth
hormone secretagogue receptor (GHS-R) (ghrelin
receptor)
3.2.1. General
characteristics of GHS-R
GHS-R belongs to the
G-protein-coupled receptors (GPCRs) that mediate extracellular to intracellular
signaling for various physiological functions. GPCRs form binding with orthosteric
or allosteric ligands that modulate their activity [37]. GHS-R, as the ghrelin receptor, mediates various
biological effects of ghrelin. Activation of GHS-R may trigger a diversity of
signaling mechanisms and physiological responses. Information on the molecular
structure of GHS-R, ligand-receptor interaction, and its intracellular
signaling pathways is essential for understanding the interaction of XNT and
GHS-R [38].
Two forms of ghrelin, AG and DAG, are primarily present in
the plasma with GOAT. DAG has antagonist properties and can counteract the
effects of AG. AG and DAG can influence the hypothalamic-pituitary-adrenal
(HPA) axis and the corticosterone/cortisol level that drives the eating desire
under stressful situations. DAG and inhibition of GOAT are good targets for
reducing obesity and bingeing-related eating disorders. Furthermore, AG/DAG ratio is an essential biomarker for
diagnosing maladaptive eating behaviors [36]. As a ligand of GHS-R, ghrelin is
considered a short-term meal initiator and a long-term energy balance
regulator. AG protein-coupled receptor is identified in the human pituitary and
hypothalamus, stimulating the GH release from the anterior pituitary.
3.2.2. Signaling
mechanism of GHS-R
There are two
GHS-R transcripts, GHS-R1a, and GHS-R1b. GHS-R1a is the acyl ghrelin receptor
that is expressed in the brain and other body areas. Multiple GHS-R1a agonists,
antagonists, and inversed agonists are available [39]. GHS-R1a can be expressed in the hypothalamus's feeding
or appetite-regulating center [24]. AG is a ligand for GHS-R1a, and acts on GHS-R1a to
stimulate GH release. The GHS-R1a is essential in eating behavior and the
pathogenetic mechanisms of drug addiction, obesity, and chronic alcohol
consumption [40].
Octanoylated ghrelin (AG) is able to activate GHS-R1a, and
is involved in multiple physiological processes, including stimulus of food
intake, gastric exhausting, body energy balance, glucose homeostasis, reduced
secretion of insulin, and lipogenesis. There are several GHS-R1a ligands. They
are peptidyl and non-peptidyl ligands that act as GHS-R1a agonists,
antagonists, or inverse agonists [41]. With their interaction, GHS-R1a
mediates the pharmacological properties of ghrelin [42]. As
a ligand of GHS-R, ghrelin may bind to GHS-R after its acylation or
octanoylation on its serine-3-residue by GOAT. Therefore, the administration of
ghrelin increases food intake and body weight. On the contrary, inhibiting its
actions with GHS-R leads to decreased food intake and weight loss.
Ghrelin
acts as an agonist at the ghrelin receptor because it modulates its maximum
efficacy and potency [43, 44]. Ghrelin is a hunger hormone that can
activate GHS-R, stimulate food intake and growth hormone secretion, and
regulate reward signaling. Therefore, ghrelin can promote body weight gain and
adipogenesis. Acylation of ghrelin at Ser3 is required for its agonistic action
on GHS-R [45]. On the contrary, inhibition of the
Ghrelin/GHS-R pathway can reduce food intake, body weight, and adiposity by
reducing appetite, increasing energy expenditure, and fat catabolism [46].
An example of an antagonist of GHS-R is liver-expressed
antimicrobial peptide-2 (LEAP-2). LEAP-2 is an endogenous non-competitive
allosteric antagonist of GHS-R1a [47]. LEAP2 as an endogenous antagonist of
GHS-R can inhibit the GHS-R activation by ghrelin and block the ghrelin’s
effects, like stimulus in food intake, GH release, and maintenance of
viable glucose levels during chronic caloric restriction [48]. LEAP-2 is recognized as an endogenous
blocker of GHS-R1a. The activity of GHS-R1a is regulated by two counter-regulatory
endogenous ligands, namely ghrelin (activation) and LEAP-2 (inhibition) [49]. LEAP-2 acts either as a competitive
ghrelin antagonist or an inverse agonist of constitutive GHS-R1a activity.
LEAP-2 can block ghrelin's effects on the stimulus of food intake and hormonal
secretion. In circulation, LEAP-2 displays an inverse activity to ghrelin, then
increases the stimulus of food intake and obesity (positive energy balance) and
decreases upon fasting and weight loss (negative energy balance). Thus, the
LEAP-2/ghrelin molar ratio varies depending on the energy status, and
modulation of this ratio conversely influences energy intake [49].
3.3.
Molecular interactions between
xanthorrhizol either with GOAT or GHS-R
3.3.1. Ligand
preparation
The canonical SMILES data of xanthorrhizol was obtained from
PubChem, and then the 3D structure of XNT was created using Marvin Sketch
software. Native ligands attached to the Ghrelin and GOAT models were separated
using Discovery Studio 2021 software. The ligands used in this study were
optimized using MOE 2019.0102 software.
3.3.2. Molecular
interaction between ghrelin and desacyl ghrelin (DAG)
Molecular docking between ghrelin and
octanoic acid; ghrelin and xanthorrhizol, GOAT protein; and XNT were performed
using the PatchDock server. The clustering RMSD parameter was set as 4 Å, and
the complex type was set as "protein-small ligand" for protein-small
ligand docking and "default" for protein-protein docking. The best
results for protein-protein docking [50]
were refined using the FIREDock server for further analysis. A higher docking score may indicate
less steric hindrance, and an ACE score may suggest a spontaneous reaction
between protein and ligand [51].
Ghrelin protein sequences with access codes NP_001289751
were obtained by searching the NCBI database using RefSeq and Homo sapiens
filter. Protein modeling of the sequence data in fasta format using the
SWISS-MODEL server (https://swissmodel.expasy.org/) [52], and the modeling results from the
SWISS-MODEL server is a ghrelin protein model with 100% similarity [53]
to the protein bank database with access code 7f9y (https://www.rcsb.org/structure/7F9Y).
Docking analysis revealed that the molecular ligand
interaction of XNT with DAG is not in the same site as octanoic acid (Fig. 4,
Table 1). Octanoic acid attaches to Ser 2, but XNT attaches to Gln 9.
3.3.3. Molecular
interaction between xanthorrhizol and GOAT
GOAT (ghrelin O-acyltransferase) protein sequences with
access codes NP_001094386.1 were obtained by searching the NCBI database using
RefSeq and Homo sapiens filter. Protein modeling of the sequence data in fasta
format using the SWISS-MODEL server (https://swissmodel.expasy.org/), and the modeling results from the
SWISS-MODEL server is a GOAT protein model with 100% similarity to the protein
bank database with access code 7f3x (https://www.rcsb.org/structure/7F3X).
Table
1.
Molecular docking prediction of Ghrelin with Octanoic Acid and Xanthorrhizol
|
No |
Compound |
Docking
Score |
Ligand
Interaction |
Type
interaction |
Distance |
|
1 |
Octanoic
Acid |
1982 |
Arg 10 |
H-acceptor |
3.23 |
|
Arg 10 |
H-acceptor |
3.04 |
|||
|
2 |
Xanthorrhizol |
3002 |
Gln 9 |
H-donor |
3.11 |
This work is licensed under the Creative Commons Attribution 4.0 License (CC BY-NC 4.0).